Defining inflammatory cell states in rheumatoid arthritis joint synovial tissues by integrating single-cell transcriptomics and mass cytometry
Fan Zhang*, Kevin Wei*, Kamil Slowikowski*, Chamith Y. Fonseka*, Deepak A. Rao*, Stephen Kelly, Susan M. Goodman, Darren Tabechian, Laura B. Hughes, Karen Salomon-Escoto, Gerald F. M. Watts, A. Helena Jonsson, Javier Rangel-Moreno, Nida Meednu, Cristina Rozo, William Apruzzese, Thomas M. Eisenhaure, David J. Lieb, David L. Boyle, Arthur M. Mandelin II, Accelerating Medicines Partnership Rheumatoid Arthritis and Systemic Lupus Erythematosus (AMP RA/SLE) Consortium, Brendan F. Boyce, Edward DiCarlo, Ellen M. Gravallese, Peter K. Gregersen, Larry Moreland, Gary S. Firestein, Nir Hacohen, Chad Nusbaum, James A. Lederer, Harris Perlman, Costantino Pitzalis, Andrew Filer, V. Michael Holers, Vivian P. Bykerk, Laura T. Donlin, Jennifer H. Anolik, Michael B. Brenner & Soumya Raychaudhuri
Nature Immunology, 2019. DOI: 10.1038/s41590-019-0378-1
View the data PDF PubMed GitHub
Abstract
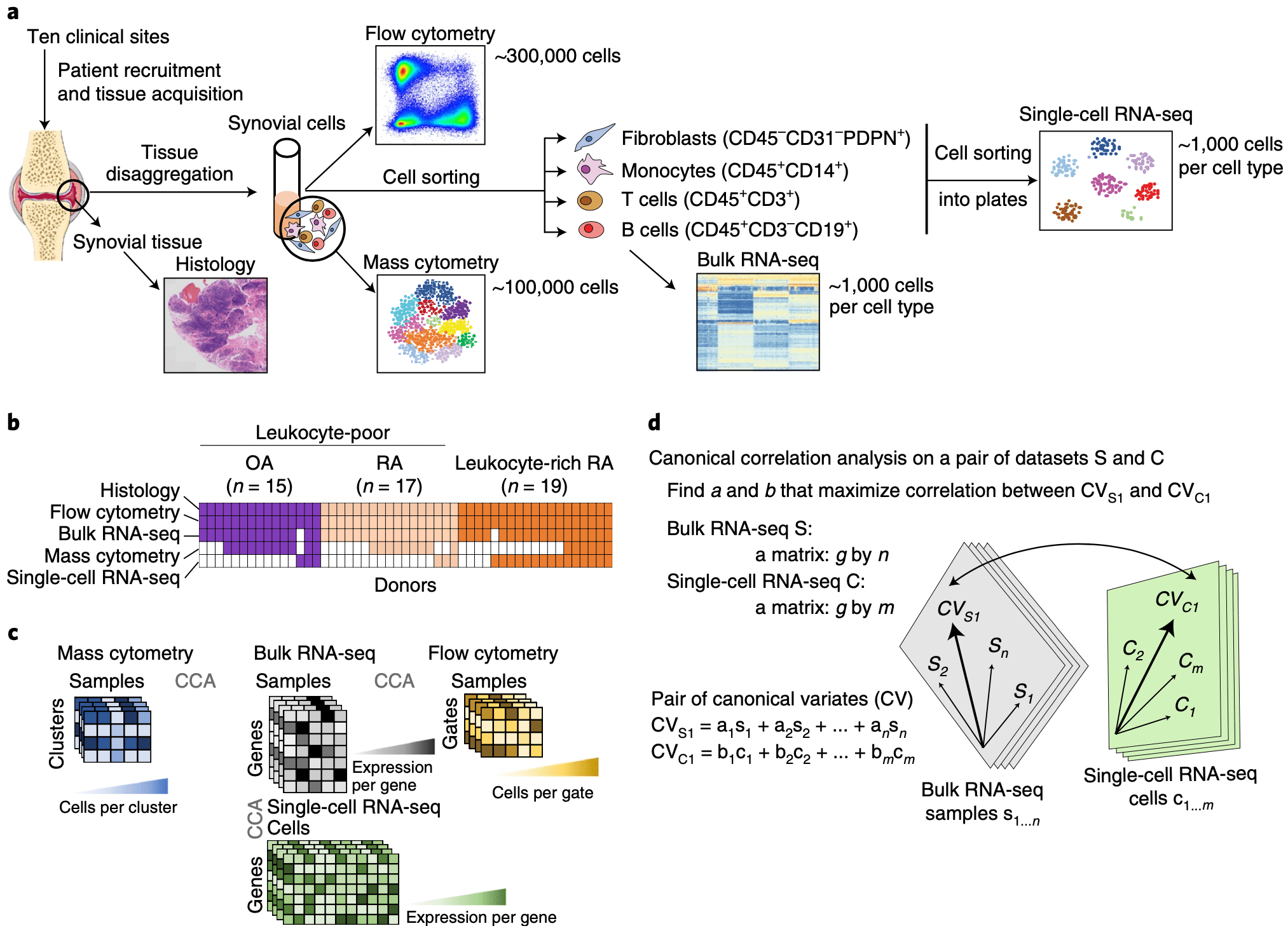
To define the cell populations that drive joint inflammation in rheumatoid arthritis (RA), we applied single-cell RNA sequencing (scRNA-seq), mass cytometry, bulk RNA sequencing (RNA-seq) and flow cytometry to T cells, B cells, monocytes, and fibroblasts from 51 samples of synovial tissue from patients with RA or osteoarthritis (OA). Utilizing an integrated strategy based on canonical correlation analysis of 5,265 scRNA-seq profiles, we identified 18 unique cell populations. Combining mass cytometry and transcriptomics revealed cell states expanded in RA synovia: THY1(CD90)+HLA-DRAhi sublining fibroblasts, IL1B+ pro-inflammatory monocytes, ITGAX+TBX21+ autoimmune-associated B cells and PDCD1+ peripheral helper T (TPH) cells and follicular helper T (TFH) cells. We defined distinct subsets of CD8+ T cells characterized by GZMK+, GZMB+, and GNLY+ phenotypes. We mapped inflammatory mediators to their source cell populations; for example, we attributed IL6 expression to THY1+HLA-DRAhi fibroblasts and IL1B production to pro-inflammatory monocytes. These populations are potentially key mediators of RA pathogenesis.